Services
Analytical services provided by A&MS-UGent
The A&MS unit is specialized in analytical method development for the determination, speciation and isotopic analysis of (ultra-)trace elements using ICP-mass spectrometry (ICP-MS). ICP-MS is considered the most powerful analytical technique for trace element analysis owing to its high sensitivity and thus, low limits of detection down to sub-ng/L, combined with multi-element capabilities, a wide linear dynamic range and high sample throughput. A&MS does not aim to compete with commercial labs for routine trace element analysis, but can be contacted for high-end applications, analytical method development, training courses and demos.
Elemental mapping of solid materials using laser ablation-ICP-MS
By combination of laser ablation (LA) for spatially resolved interrogation of solid materials and ICP-MS for elemental analysis, the quantitative 2-dimensional distribution of elements can be revealed in materials such as biological tissue, rocks, minerals, polymers, etc. The approach is characterized by a spatial resolution of 100 µm down to ± 1 µm and offers LODs down to the sub-ng/g level. Due to hardware developments realized by A&MS-UGent, elemental mapping can be performed at unprecedented speed.1,2

Figure 1. Schematic representation of laser ablation-ICP-MS with 3 elemental mapping examples: (1) high-resolution slide scanner image of H&E-stained kidney tissue and corresponding high-resolution Pt distribution map obtained via LA-ICP-MS (1 µm spatial resolution). (2) Multi-elemental RGB (Na, P and Fe) LA-ICP-MS map of a mouse embryo (20 µm spatial resolution) (3) Optical image and RGB (Ca, Si and Fe) LA-ICP-MS map of meteorite Y-790960 (5 µm spatial resolution).3-5
Single-particle and single-cell ICP-MS
By using a fast-scanning ICP-MS unit, engineered nanoparticles (ENPs) can be characterized on an individual basis, thus providing information on the number and mass concentration in an aqueous suspension, as well as on their size distribution. In a similar approach, also the content of both exogenous and endogenous elements can be determined in individual bacterial, plant, animal and human cells, down to femtogram levels. As a result, not only the average content is obtained, but also the spread in content across a cell population is revealed.
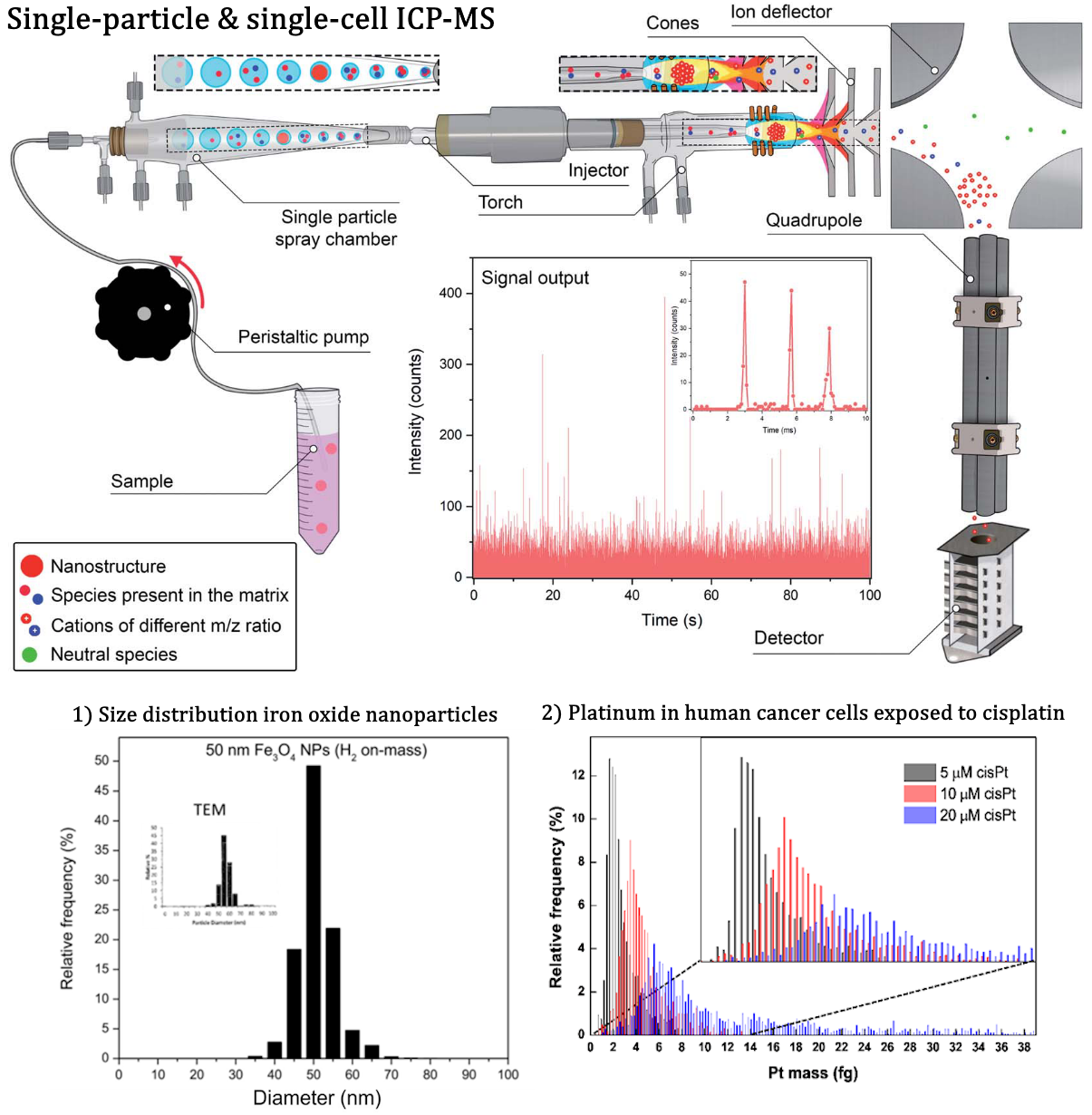
Figure 2. Schematic representation of single-particle & single-cell ICP-MS with 2 examples: (1) histogram displaying the size distribution of iron oxide nanoparticles.6 (2) Pt content in individual human cancer cells upon exposure to cisplatin at various concentrations.7
High-precision isotopic analysis via multi-collector ICP-MS
The A&MS unit houses 2 multi-collector ICP-MS (MC-ICP-MS) units dedicated to isotopic analysis of metals, metalloids and some metals at ultra-high precision (down to 0.001% SD) and is equipped with a class-10 clean lab for contamination-free sample preparation. Isotopic analysis of Sr is, e.g., used for provenance determination of foodstuff and human remains, isotopic analysis of Pb to discriminate between crustal lead and anthropogenic contamination and for provenance determination of objects of art. Isotopic analysis of Hg in environmental samples provides insight into its sources. Other applications rely on the isotopic composition of, among other, B, Cu, Fe, K, Li, Mg, Nd, S and Zn.
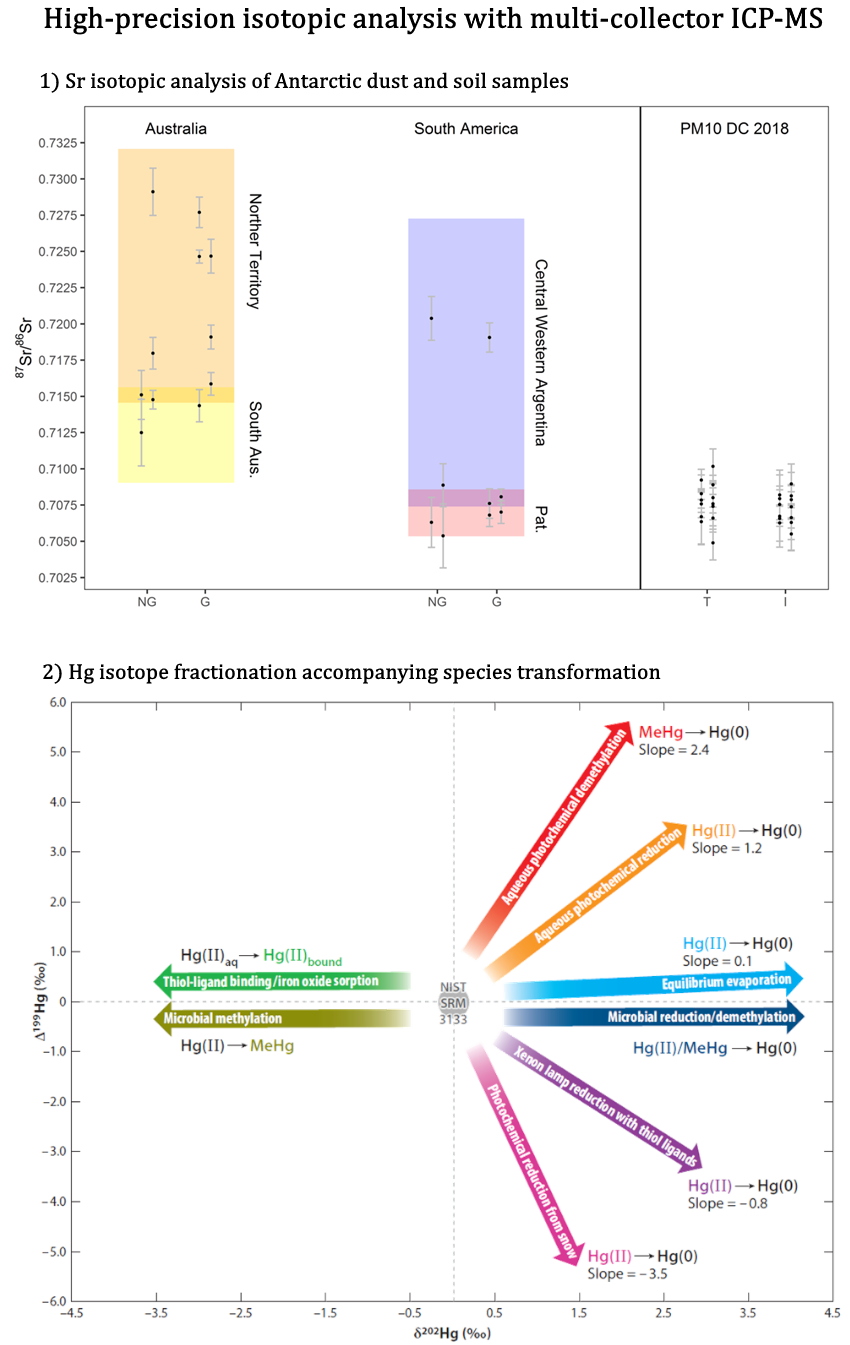
Figure 3. Two examples of high-precision isotopic analysis via MC-ICP-MS: (1) 87Sr/86Sr isotope ratio in Antarctic dust (PM10 fraction) and soil samples of potential source areas8 and (2) Hg isotope fractionation accompanying species transformations.9
For more information and/or a quote, please contact: frank.vanhaecke@ugent.be or thibaut.vanacker@ugent.be.
References
1. T. Van Acker, S. J. M. Van Malderen, T. Van Helden, C. Stremtan, M. Šala, J. T. van Elteren and F. Vanhaecke, J. Anal. At. Spectrom., 2021, 36, 1201–1209.
2. S. J. M. Van Malderen, T. Van Acker and F. Vanhaecke, Anal. Chem., 2020, 92, 5756–5764.
3. A. Gundlach-Graham and D. Günther, Anal. Bioanal. Chem., 2016, 408, 2687–2695.
4. T. Van Acker, S. J. M. Van Malderen, M. Van Heerden, J. E. McDuffie, F. Cuyckens and F. Vanhaecke, Anal. Chim. Acta, 2016, 945, 23–30.
5. R. Maeda, T. Van Acker, F. Vanhaecke, A. Yamaguchi, V. Debaille, P. Claeys and S. Goderis, J. Anal. At. Spectrom., 2023, 38, 369–381.
6. E. Bolea-Fernandez, D. Leite, A. Rua-Ibarz, T. Liu, G. Woods, M. Aramendia, M. Resano and F. Vanhaecke, Anal. Chim. Acta, 2019, 1077, 95–106.
7. T. Liu, E. Bolea-Fernandez, C. Mangodt, O. De Wever and F. Vanhaecke, Anal. Chim. Acta, 2021, 1177, 338797.
8. S. Bertinetti, E. Bolea-Fernandez, M. Malandrino, B. Moroni, D. Cappelletti, M. Grotti and F. Vanhaecke, J. Anal. At. Spectrom., 2022, 37, 103–113.
9. J. D. Blum, L. S. Sherman and M. W. Johnson, Annu. Rev. Earth Planet. Sci., 2014, 42, 249–269.